octacalcium phosphate, Ca 8H 2(PO 4) 6.whitlockite, Ca 9(Mg,Fe)(PO 4) 6(PO 3OH). 
The mineral proportion of calculus ranges from approximately 40–60%, depending on its location in the dentition, and consists primarily of calcium phosphate crystals organized into four principal mineral phases, listed here in order of decreasing ratio of phosphate to calcium: Calculus chemical composition Ĭalculus is composed of both inorganic (mineral) and organic (cellular and extracellular matrix) components. This came to be a term used for calcium phosphate on teeth in the early 19th century. potassium bitartrate, commonly known as cream of tartar). Tartar, on the other hand, originates in Greek as well ( tartaron), but as the term for the white encrustation inside casks (a.k.a. 
This spun off many modern words, including "calculate" (use stones for mathematical purposes), and "calculus", which came to be used, in the 18th century, for accidental or incidental mineral buildups in human and animal bodies, like kidney stones and minerals on teeth. Calculus was a term used for various kinds of stones. The word comes from Latin calculus "small stone", from calx "limestone, lime", probably related to Greek χάλιξ chalix "small stone, pebble, rubble", which many trace to a Proto-Indo-European root for "split, break up". Calculus buildup can be removed with ultrasonic tools or dental hand instruments (such as a periodontal scaler). Brushing and flossing can remove plaque from which calculus forms however, once formed, calculus is too hard (firmly attached) to be removed with a toothbrush. Calculus can form both along the gumline, where it is referred to as supragingival ("above the gum"), and within the narrow sulcus that exists between the teeth and the gingiva, where it is referred to as subgingival ("below the gum").Ĭalculus formation is associated with a number of clinical manifestations, including bad breath, receding gums and chronically inflamed gingiva. This leads to calculus buildup, which compromises the health of the gingiva (gums). 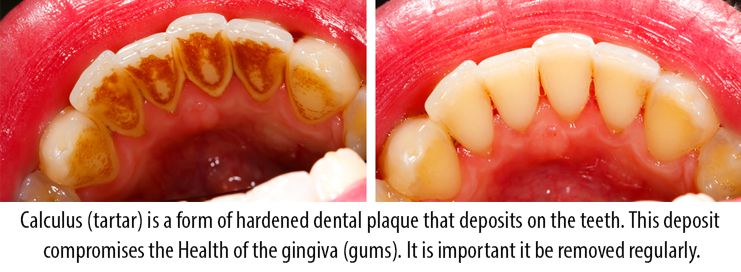
This process of precipitation kills the bacterial cells within dental plaque, but the rough and hardened surface that is formed provides an ideal surface for further plaque formation. It is caused by precipitation of minerals from saliva and gingival crevicular fluid (GCF) in plaque on the teeth. In dentistry, calculus or tartar is a form of hardened dental plaque. Heavy staining and calculus deposits exhibited on the lingual surface of the mandibular anterior teeth, along the gumline Calculus deposit (indicated with a red arrow) on x-ray image For other uses, see Calculus (disambiguation). Because young children may swallow toothpaste when brushing, which can lead to fluorosis, they should use children's toothpastes, which contain lower amounts of fluoride.This article is about the dental problem. Also, fluoridated toothpaste should be used by people of all ages. read more, causing mild to severe discoloration of teeth. The main source of fluoride is fluoridated drinking water. Fluoride (the ionic form of fluorine) is widely distributed in nature. Excessive fluoride exposure may cause dental fluorosis Fluorine Toxicity Most of the body’s fluorine (F) is contained in bones and teeth. The dose must be selected according to the amount of fluoride present in the drinking water and diet, the age of the child, and whether topical fluoride is being used in toothpaste and/or applied during dental care. Tablets should be swished in the mouth to enhance topical fluoride absorption before swallowing. Oral supplements for children are available in toothpastes, liquids (for use in a dropper for infants), gels, and tablets.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
